Does aldol condensation occur in glycolysis?
Aldol and retro-aldol are two important classes of chemical reactions, occasionally encountered in biochemistry too. The cleavage of the 1,6 phosphate diester of fructose in glycolysis (sugar burning) pathway is an example of a retro-aldol reaction.
What is retro-aldol reaction?
Retro-aldol reaction: A reaction in which a β-hydroxy carbonyl compound decomposes into an aldehyde or ketone, plus another carbonyl compound. The formal reverse of the aldol reaction.
What is the disadvantage of aldol reaction?
The reaction conditions used for the aldol condensation of 3-pentanone described above are rather harsh, and this is a disadvantage if elimination of water from the aldol product occurs, or if self-condensation occurs under these equilibrating conditions.
What does an aldol reaction produce?
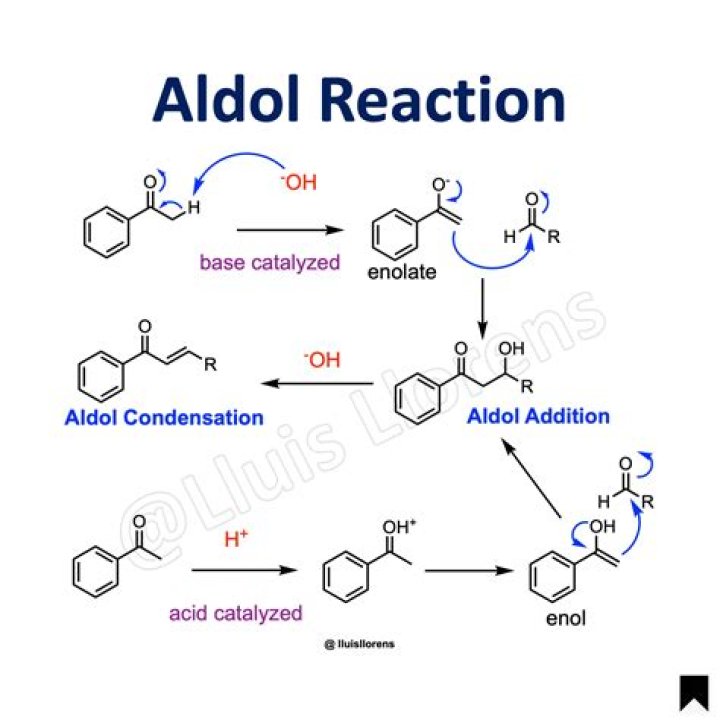
The aldol reaction is a means of forming carbon–carbon bonds in organic chemistry. These products are known as aldols, from the aldehyde + alcohol, a structural motif seen in many of the products. Aldol structural units are found in many important molecules, whether naturally occurring or synthetic.
Is glucose reduced or oxidized in glycolysis?
Catabolic pathway during which a 6 carbon glucose molecule is split into two 3 carbon sugars which are then oxidized and rearranged by a step-wise metabolic process that produces two molecules of pyruvic acid. The reactions of glycolysis occur in 2 phases: 1.
Which Carbohydrateanion is formed in aldol condensation?
Aldol Condensation can be defined as an organic reaction in which enolate ion reacts with a carbonyl compound to form β-hydroxy ketone or β-hydroxy aldehyde, followed by dehydration to give a conjugated enone.
What is claisen Schmidt reaction?
The Claisen-Schmidt condensation reaction is an organic reaction in which a ketone or an aldehyde holding an α-hydrogen reacts with an aromatic carbonyl compound which does not have any α-hydrogens. This reaction is named after the chemists J.G. Schmidt and Rainer Ludwig Claisen.
Which of the following is reduced by sodium borohydride?
Sodium borohydride is a relatively selective reducing agent. Ethanolic solutions of sodium borohydride reduce aldehydes and ketones in the presence of epoxides, esters, lactones, acids, nitriles, or nitro groups. Reduction of aldehydes is straightforward.
How can you minimize the number of possible products in an aldol reaction?
1) A reaction of an aldehyde with no alpha-hydrogens with a ketone that has alpha hydrogens: Aldehydes lacking alpha-hydrogens cannot form an enolate so they can only function as electrophilic acceptor reactants. This reduces the number of possible products by half.
Which molecule is reduced in glycolysis?
NAD+ is the oxidized form of the molecule; NADH is the reduced form of the molecule after it has accepted two electrons and a proton (which together are the equivalent of a hydrogen atom with an extra electron).
What is the most oxidized compound in glycolysis?
In most cells glycolysis converts glucose to pyruvate which is subsequently oxidized to carbon dioxide and water by mitochondrial enzymes. Obligate ATP production via glycolysis also occurs in the absence of oxygen whether mitochondria are present or not.
What is carbonyl condensation?
A carbonyl condensation reaction takes place between two carbonyl partners and involves both. nucleophilic addition and. α-substitution. One carbonyl is converted by base into a nucleophilic enolate ion, which then adds to the electrophilic. carbonyl group of the second compound.