Can you produce methanol from CO2?
The methanol synthesis technology from CO2 using heterogeneous catalysts has reached relatively high level of maturity corresponding to the demonstration units. A demonstration plant in Iceland is already producing annually 4000 tons of methanol from CO2.
How is methanol made from CO2 and H2?
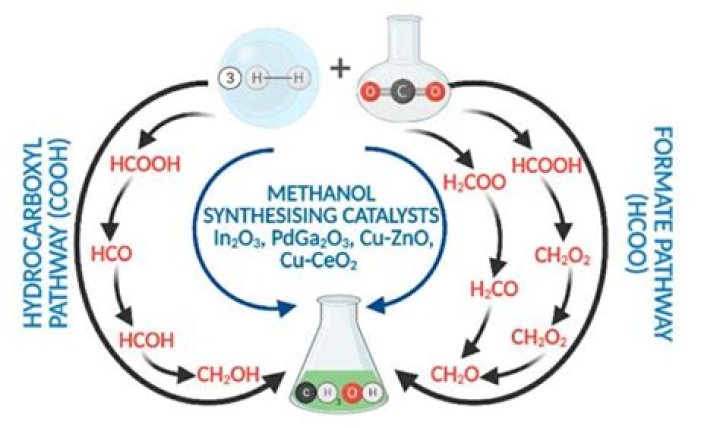
Alternative to syngas, methanol can be produced by directly hydrogenating pure CO2 with H2 with high selectivity on conventional Cu/ZnO-based catalysts. However, the reaction rates are lower than with syngas feeds [9]. The equilibrium conversions are also lower compared to CO hydrogenation [10].
How is converted into methanol?
The carbon dioxide hydrogenation process works by decomposing water to create a hydrogen gas using renewable energy, which then bonds with the carbon dioxide on the surface of the catalyst to create methanol. The process essentially converts greenhouse gases to fuels that emit carbon dioxide when burned.
How do you turn methanol into gasoline?
The methanol is obtained by steam reforming natural gas to synthesis gas. In the process, methanol is first converted to dimethyl ether by being passed over a fixed bed gamma-alumina at pressures of 15–175 psig. The conversion is very exothermic with 750 BTUs of heat produced per pound of methanol.
How is CO2 converted into ethanol?
Carbon + copper = magic. A new catalyst turns carbon dioxide into ethanol at over 90 percent efficiency. Separating chemical elements can be complex and expensive because of strong bonds. This catalyst is an electrified arrangement of copper on a structure of carbon.
How do you make methanol synthesis?
Methanol synthesis is considered via the direct CO2 hydrogenation process concept proposed by Bongartz et al. (2019) (Figure 2). AA-grade methanol is produced at a reaction pressure of 70 bar, a reaction temperature of 250° C, a molar H2:CO2 ratio of 3.0 and application of commercial Cu/ZnO/Al2O3 catalyst.
Why is methanol a good solvent?
Methanol, CH3OH, is a good solvent for polar substances because: (1) Methanol undergoes dipole-dipole interactions. (2) Methanol is able to form hydrogen bonds with the polar substances. (3) There are weak ion-dipole interactions in methanol. (4) The van der Waals forces in methanol cause repulsion.
Is methanol a solvent?
Methanol is primarily used as an industrial solvent to help create inks, resins, adhesives, and dyes. It is also used as a solvent in the manufacture of important pharmaceutical ingredients and products such as cholesterol, streptomycin, vitamins and hormones.
Does methanol mix with gasoline?
As is common for most alcohols used in gasoline, methanol is fully soluble in water and also miscible with gasoline-type hydrocarbons. In fact, methanol gasoline blends have cleaner burning properties that generally reduces CO, hydrocarbon (HC), PM and other pollutants from most gasoline engine vehicles.